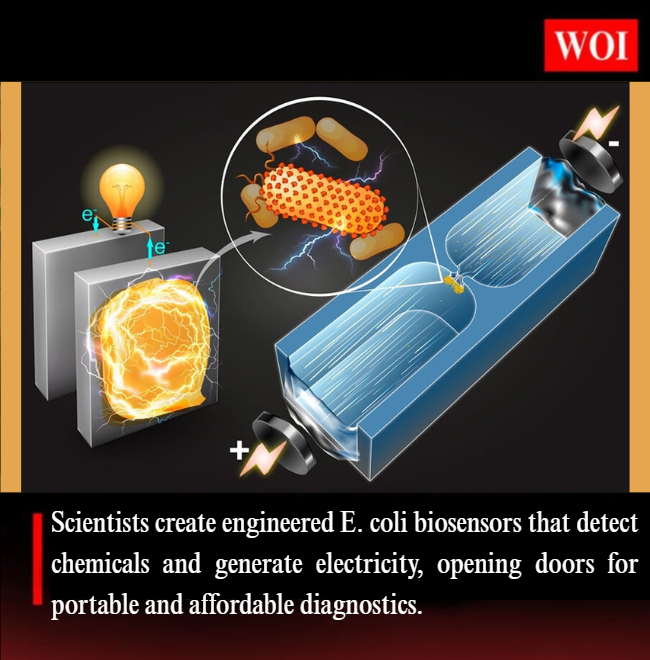
Recent breakthroughs in synthetic biology and bioelectrochemistry have led to the creation of self-powered living biosensors using genetically engineered Escherichia coli (E. coli). Developed by researchers at Imperial College London and Zhejiang University, these biosensors convert chemical signals directly into electrical outputs, paving the way for low-cost, portable, and programmable bioelectronic devices.
Why Living Biosensors?
Traditional enzyme-based biosensors are fragile, costly, and slow in complex environments. Whole-cell biosensors often rely on optical signals that are difficult to integrate with portable electronics. The new approach overcomes these hurdles by engineering bacteria that can detect chemicals and generate electrical currents.
How They Work
The modified E. coli contain three modules:
- Sensing: Detects target chemicals like sugars or heavy metals.
- Processing: Amplifies the detected signal.
- Output: Produces phenazines, molecules measurable via electrochemical methods.
Two biosensors were demonstrated:
- Detection of arabinose (plant sugar) within 2 hours.
- Detection of mercury ions in water at levels below WHO safety limits within 3 hours.
Researchers also built an ‘AND’ logic gate inside E. coli, showing that living sensors can perform basic biochemical computing.
Applications
These self-sustaining biosensors can work in contaminated environments and link easily with portable electronics. Potential applications include environmental monitoring, medical diagnostics, and food safety testing.
About E. coli
Most E. coli strains are harmless gut bacteria, but some, like E. coli O157:H7, cause foodborne illnesses. They survive in acidic foods and require proper cooking (≥70 °C) for safe consumption.
This innovation combines biology and electronics, offering a future where living systems become powerful tools for real-time detection and diagnostics.